
PIPELINE
R&D APPROACH
CONNECTA follows a network pharmacology-based approach by evaluating all the targets involved in CNS properties and processes, and modulating different pathways that lead to specific disorders.
The traditional one drug–one target–one disease approach in CNS drug discovery has become increasingly inefficient. CONNECTA’s integrative in silico approach is based on identifying multiple actionable targets and designing drug candidates with disease-modifying potential.
THERAPEUTIC FOCUS
CONNECTA is currently focused on the clinical development of new treatments for patients suffering from neurodevelopmental disorders with abnormalities of protein networks involved in synaptic and structural plasticity (neuroplasticity imbalance).
PIPELINE
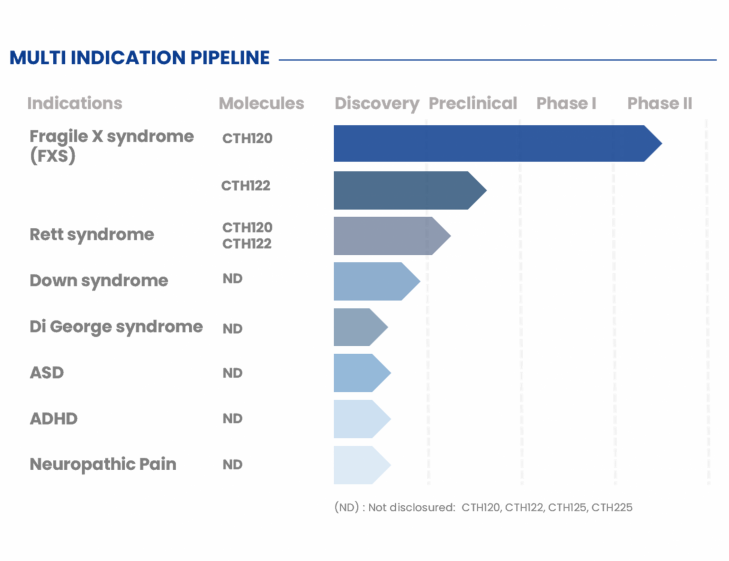
CTH120 – NEUROPLASTICITY MODULATOR
CONNECTA’s leading program, CTH120, is a first-in-class small molecule, NCE, for neuroplasticity-mediated disorders with social behaviour and pro-cognitive properties.
CTH120 was designed after the study of more than 30 molecular and cellular targets for neuroplasticity. The molecule has proven to be, in in vitro tests and in vivo in animal models, an excellent neuroplasticity modulator with a great extension profile for different neurodevelopmental disorders (fragile X syndrome, Rett syndrome, Down syndrome and autism spectrum disorders, among others).
Excellent preclinical efficacy results: CTH120 rescues the dendritic pathology of fragile X syndrome (FXS), clearly improving cognitive ability and completely restoring the social abnormalities linked to the disorder.
Novel mechanism of action: CTH120 modulates neuroplasticity by acting on neurotrophic factors pathways.
International patent family.
Orphan drug designation: The EMA has granted orphan drug status for CTH120 in FXS, as orphan medicinal product nr EU/3/21/2432 (May 2021).
Completed FIH studies: CTH120 has completed Phase I clinical trials in Q4 2024, with positive results.
Entering Phase II clinical trials: CTH120 has completed enabling Phase II non clinical studies, and it is ready to submit the Clinical Trial Application for the assessment of safety, tolerability and efficacy of CTH120 in FXS adult subjects.
CTH122 – NEUROPLASTICITY MODULATOR
CTH122 is a recently patented First-in-class small molecule with neuroplasticity modulation properties and demonstrated efficacy in FXS and Rett syndrome preclinical models.
ABOUT FRAGILE X SYNDROME
Fragile X syndrome (FXS) is a genetic condition that causes a range of developmental problems including learning disabilities and cognitive impairment. FXS’s estimated prevalence for male and female patients is approximately 3/10.000.
Affected individuals usually exhibit delayed development of speech and language by age 2. Most males with FXS have mild to moderate intellectual disability, while about one-third of affected females are intellectually disabled. Children with FXS may also have anxiety and hyperactive behavior such as fidgeting or impulsive actions. They may have attention deficit disorder (ADD), which includes an impaired ability to maintain attention and difficulty focusing on specific tasks. About one-third of individuals with FXS have features of autism spectrum disorder that affect communication and social interaction. Seizures occur in about 15% of males and about 5% of females with FXS.
Source: https://ghr.nlm.nih.gov/condition/fragile-x-syndrome



